
AI in Interventional Radiology & Cardiology
Artificial intelligence (AI) is transforming interventional cardiology and radiology by advancing image interpretation, streamlining procedural workflows, and lowering radiation exposure. Deep learning–based image analysis supports automated detection of coronary lesions, assessment of plaque characteristics, and real-time procedural guidance, enhancing diagnostic accuracy and reproducibility. In robotic-assisted interventions, AI-integrated systems improve precision, facilitate automated catheter navigation, and reduce operator fatigue, contributing to safer and more standardized procedures.
Read More
Innovations in Interventional Cardiology: Advancing Minimally Invasive Cardiac Care
Recent advances in interventional cardiology have transformed minimally invasive strategies for the diagnosis and treatment of cardiovascular disease. Technologies such as transcatheter aortic valve replacement (TAVR), mitral transcatheter edge-to-edge repair (MTEER), tricuspid repair (TTEER) and replacement (TTVR) systems, and bioresorbable stents are reducing dependence on open-heart surgery, lowering procedural risk, and enhancing recovery. Advanced imaging modalities, including intravascular ultrasound (IVUS) and optical coherence tomography (OCT), provide precise procedural guidance, while robotic-assisted platforms improve operator accuracy. Collectively, these innovations have broadened therapeutic options and enabled management of complex coronary and structural heart disease.
Read More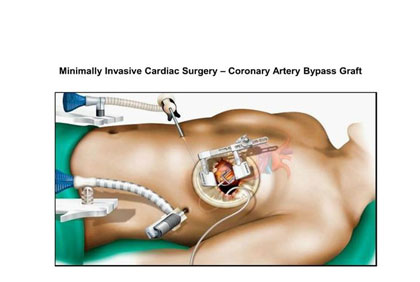
World-First Coronary Bypass without Opening the Chest
In a world-first procedure, researchers at the National Institutes of Health and Emory School of Medicine performed a coronary artery bypass without cutting the chest wall. The team used a novel intervention to prevent coronary artery blockage — a rare but often fatal complication after heart-valve replacement. This approach may offer a safer, less traumatic alternative to open-heart surgery for high-risk patients at risk of coronary artery obstruction.
Read More
Abluminus DES+ vs XIENCE — ABILITY (diabetes) 12-month outcomes
In patients with diabetes undergoing PCI, the Abluminus DES+ sirolimus-eluting stent showed higher 12-month ischemia-driven TLR and target-lesion failure than XIENCE EES — raises questions about platform performance in high-risk metabolic patients.
Read More
Late-Breaking Analysis Shows Similar Long-Term Survival with Two Multi-Arterial CABG Approaches
A late-breaking study utilizing over 15 years of national outcomes data from the STS Adult Cardiac Surgery Database (ACSD) indicates that the two most frequently used multi-arterial coronary artery bypass grafting (CABG) strategies—bilateral internal thoracic artery (BITA) and single internal thoracic artery plus radial artery (SITA+RA)—demonstrate comparable overall long-term survival, with notable variations observed according to patient age.
Read More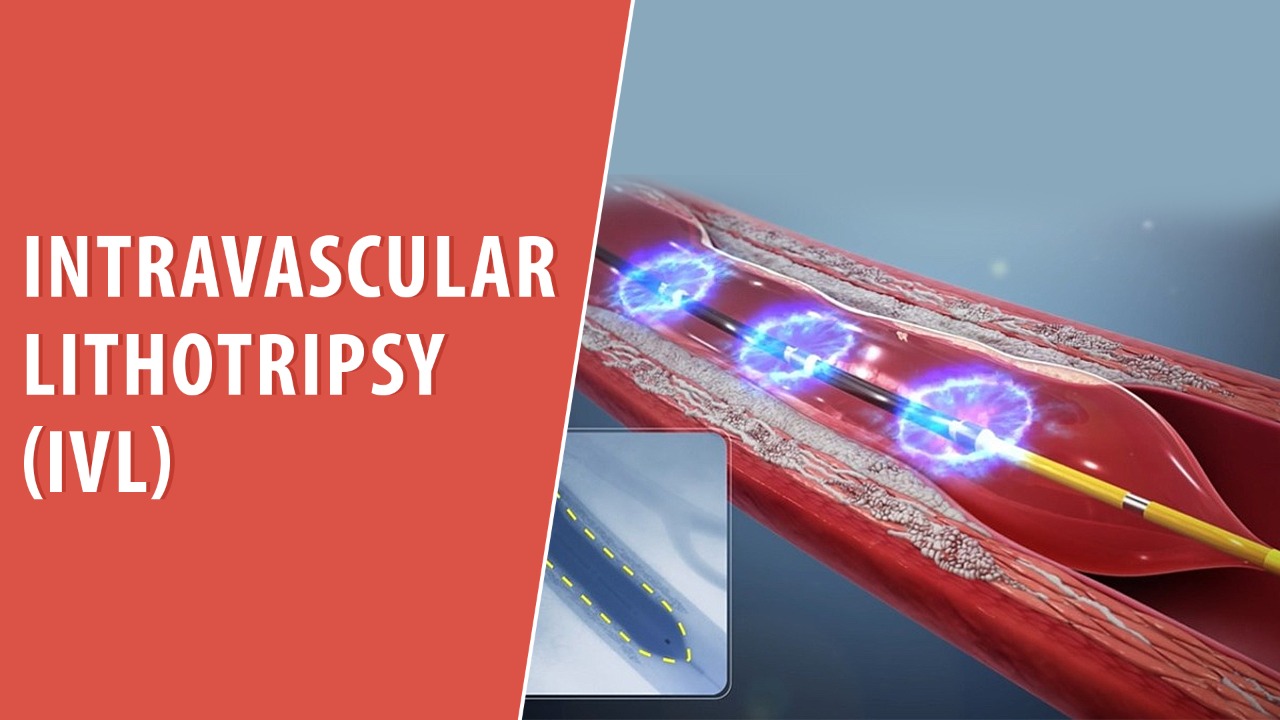
Intravascular lithotripsy (IVL) — expanding role for calcified coronaries
IVL continues to gain momentum as a safe, effective method to fracture deep coronary calcium and facilitate optimal stent expansion in heavily calcified lesions; longer-term outcomes and broader lesion-set validation are ongoing priorities.
Read More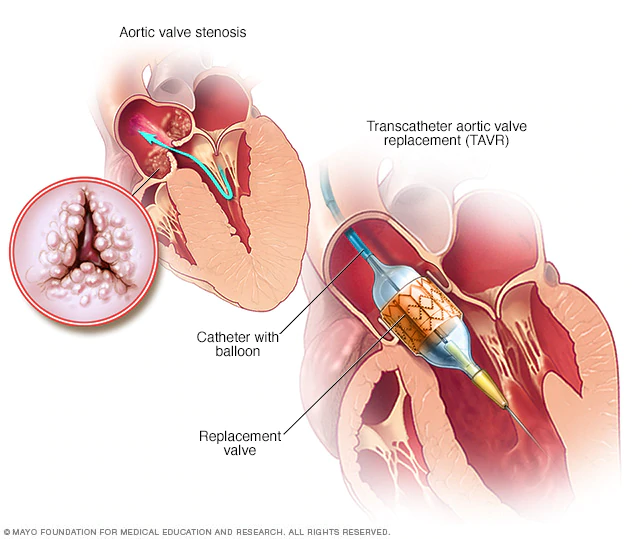
TAVR Program Standards and Policy Impact
The Societies recommend that CMS revise the TAVR NCD to incorporate newly FDA-approved indications, including treatment of asymptomatic aortic stenosis, and to anticipate future expanded indications such as aortic regurgitation, bicuspid aortic stenosis, and moderate aortic stenosis. As TAVR has become central to aortic stenosis management, an updated NCD is expected to have broad implications, affecting both patients undergoing TAVR and the overall care systems for aortic valve disease.
Read More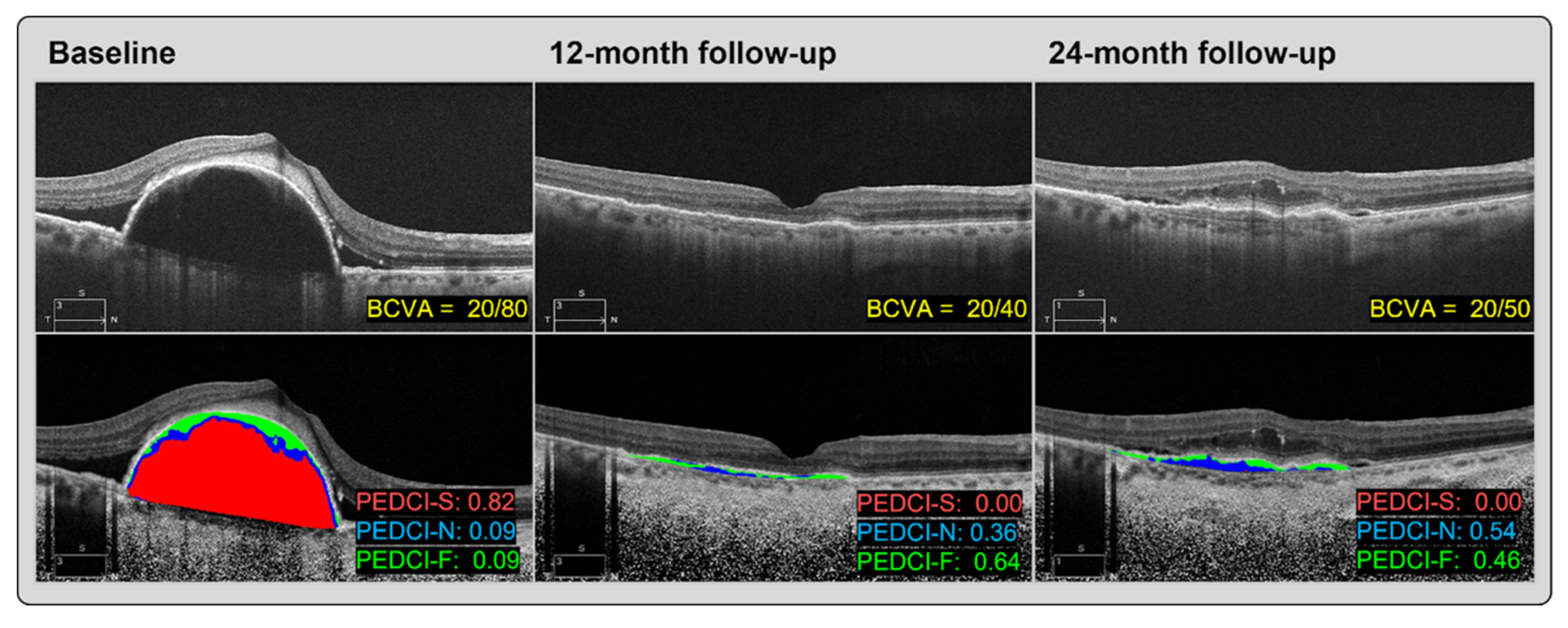
Optical coherence tomography (OCT) prognostic studies — 2026 imaging outcomes
New 2026 OCT prognostic data and trials emphasize the role of intravascular imaging in predicting outcomes and optimizing stent deployment to reduce late events.
Read More
Atherectomy strategy updates — rotational vs orbital insights
The Task Force on Rotational Atherectomy of the Japanese Association of Cardiovascular Intervention and Therapeutics (CVIT) first issued an expert consensus document in 2020 summarizing techniques and evidence for rotational atherectomy (RA), with an update in 2023. Contemporary 2026 expert documents and trial follow-ups highlight RA vs OA technique nuances; RA produced larger stent expansion in some OCT-guided comparisons, but device selection remains lesion-specific.
Read More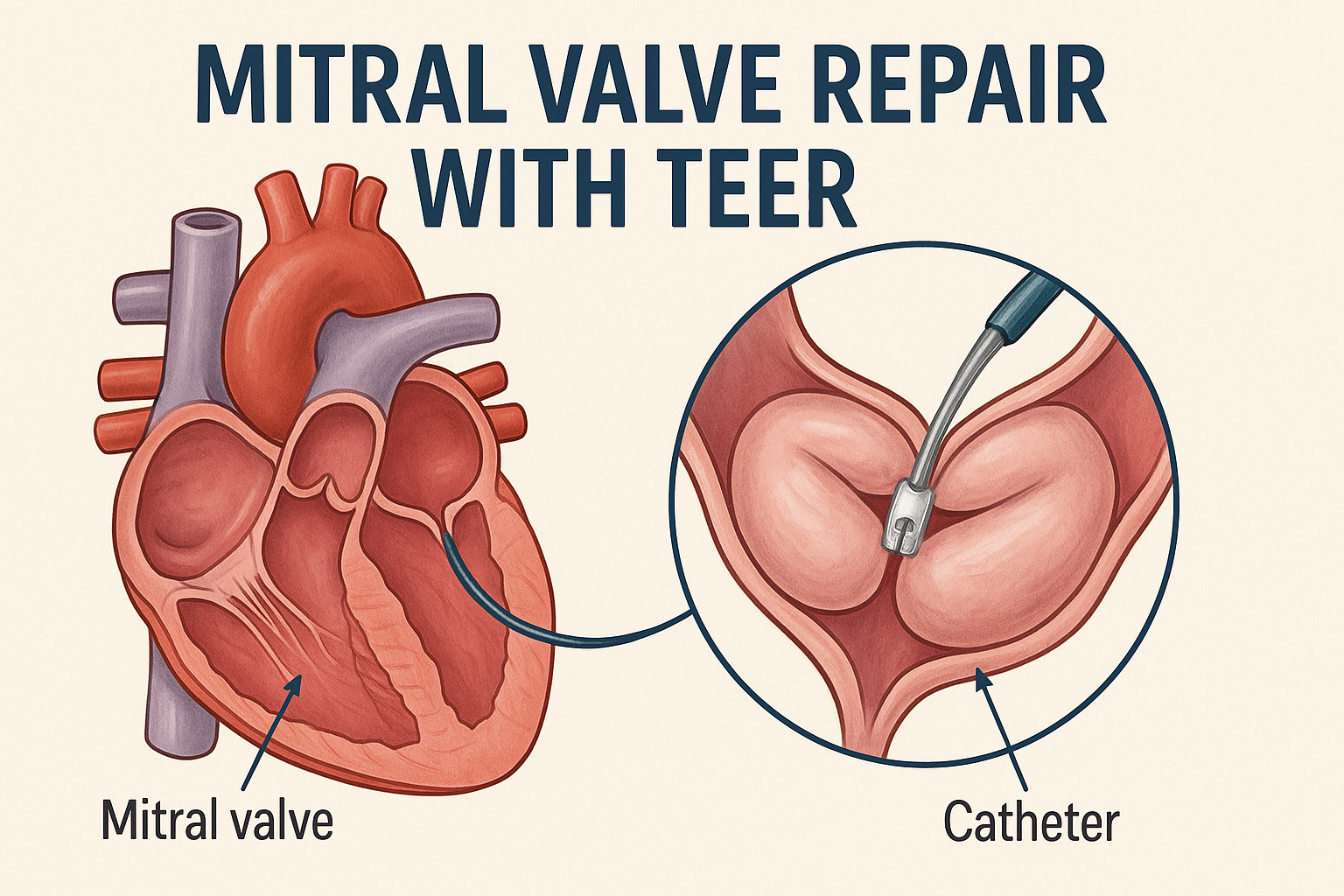
Real-World Comparison: TEER vs Surgical Repair for Mitral Regurgitation
In a large real-world cohort, transcatheter edge-to-edge repair (TEER) and surgical repair for mitral regurgitation (MR) demonstrated differing outcomes. Early mortality was comparable; however, surgery was associated with improved survival beyond two months. TEER was linked to fewer early episodes of dyspnea, atrial fibrillation, and heart failure. Rates of major adverse cardiovascular events (MACE), stroke, acute myocardial infarction (AMI), and emergency department visits were similar, supporting individualized treatment selection for MR.
Read More
Medtronic to Acquire CathWorks
Medtronic has confirmed it will exercise its option to acquire CathWorks, a privately held medical device company specializing in the diagnosis and treatment of coronary artery disease (CAD) through its fractional flow reserve (FFR) diagnostic technology.
Read More
ALPHA-PE Research Fund Introduced to Enhance Pulmonary Embolism Care
AngioDynamics, Inc. and The PERT Consortium have announced the establishment of the ALPHA-PE Research Fund, an investigator-initiated program aimed at supporting independent research in pulmonary embolism (PE). According to the press release, ALPHA-PE is intended to promote physician-led studies that address key evidence gaps in PE management, an area frequently challenged by limited real-world data to guide clinical decision-making.
Read More
Nipro’s Avantec Vascular Secures FDA Clearance for DVT Thrombectomy System
Avantec Vascular, a Nipro company, announced that it has received FDA 510(k) clearance for its thrombectomy system indicated for the treatment of deep vein thrombosis (DVT). The device is specifically cleared for the removal of fresh, soft emboli and thrombi from peripheral venous vessels measuring ≥ 7 mm in diameter.
Read More
Robocath Initiates First-in-Human Study of Second-Generation Robotic System for CAD
Robocath, a France-based developer of robotic technologies for interventional cardiology, announced the initiation of a first-in-human (FIH) clinical study assessing its latest robotic system designed for the treatment of coronary artery disease (CAD). According to the company, the second-generation platform incorporates enhanced capabilities aimed at addressing the growing complexity of contemporary coronary interventions.
Read More
Burl’s Foqus Ultrasound System Utilized in First-in-Human PFO Detection Cases
Burl Concepts, a company developing diagnostic technology to assess cerebral blood flow and detect emboli, announced the initial human applications of its Foqus ultrasound system for the identification of brain emboli. The system was employed to detect patent foramen ovale (PFO) in 30 patients across the Netherlands and the United Kingdom.
Read More
HighLife TMVR System Granted CE Mark Approval
HighLife SAS, a France-based company developing transcatheter therapies for structural heart disease, announced that its HighLife transcatheter mitral valve replacement (TMVR) system has received CE Mark approval.
Read More
Concept’s Abluminus NP Polymer-Free DES and Antiplatelet Approaches Evaluated in STARS-DAPT
Concept Medical Inc. announced the enrollment of the first patients in the STARS-DAPT randomized controlled trial (RCT), involving individuals with ST-segment elevation myocardial infarction (STEMI) treated with the company’s Abluminus NP—a polymer-free, sirolimus-based, nanocarrier-eluting stent. The study is assessing a P2Y12 inhibitor–based single antiplatelet regimen following a short course of dual antiplatelet therapy (DAPT), compared with conventional DAPT.
Read More
CVRx Launches BENEFIT-HF Trial Evaluating Barostim in a Broader Heart Failure Population
CVRx, Inc., a company focused on neuromodulation therapies for cardiovascular disease, announced the initiation of the BENEFIT-HF clinical trial. According to the company, this randomized study is designed to assess the effects of its Barostim neuromodulation device in an expanded population of patients with heart failure.
Read More
Boston Scientific to Acquire Penumbra
Boston Scientific Corporation and Penumbra, Inc. have entered into a definitive agreement under which Boston Scientific will acquire Penumbra in a cash-and-stock transaction valuing the company at $374 per share, representing an enterprise value of approximately $14.5 billion. Penumbra’s portfolio includes devices for the treatment of pulmonary embolism, stroke, deep vein thrombosis, acute limb ischemia, myocardial infarction, and aneurysms.
Read More
Rampart Initiates Multispecialty SECURE Clinical Program for Radiation Protection
Rampart, an Alabama-based developer of barrier-based radiation protection technologies, announced the launch of the SECURE clinical program, a series of studies evaluating the clinical utility of radiation mitigation equipment. The coordinated program spans multiple interventional specialties—including interventional cardiology, electrophysiology, endovascular, aortic, and neurovascular—and is assessing the use of Rampart’s enhanced radiation protection devices (ERPDs). According to the company, the initiative aims to expand understanding of how contemporary radiation protection strategies can enhance safety for clinicians and care teams without compromising procedural performance.
Read More
ACC/AHA Release Updated Guideline on Adult Congenital Heart Disease Management
The American College of Cardiology and the American Heart Association have issued a new guideline for the management of congenital heart disease in adults. The document was developed in collaboration with and endorsed by the Heart Rhythm Society, the International Society for Adult Congenital Heart Disease, and the Society for Cardiovascular Angiography and Interventions. According to the ACC/AHA press release, the guideline offers updated recommendations for surveillance, counseling, and treatment of adults with congenital heart disease, incorporating evidence published between 2017 and 2024 and replacing the 2018 guidance.
Read More